In a groundbreaking study published online in Cell on April 3, 2025, researchers led by BGI-Research along with partners from the Center for Excellence in Brain Science and Intelligence Technology at Chinese Academy of Sciences, Centre de Recherche INSERM, Lingang Laboratory, Tencent AI for Life Sciences Lab, and other leading institutions have presented the first single-cell spatial transcriptome atlas of the macaque claustrum. By integrating cutting-edge single-nucleus RNA sequencing and high-resolution spatial transcriptomics with comprehensive whole-brain connectivity mapping, the team has provided a panoramic view of the molecular and circuit architecture of this elusive brain region, which has long been implicated in consciousness and cognitive processing.
 The study “Single-cell spatial transcriptome atlas and whole-brain connectivity of the macaque claustrum” was published in Cell.
The study “Single-cell spatial transcriptome atlas and whole-brain connectivity of the macaque claustrum” was published in Cell.
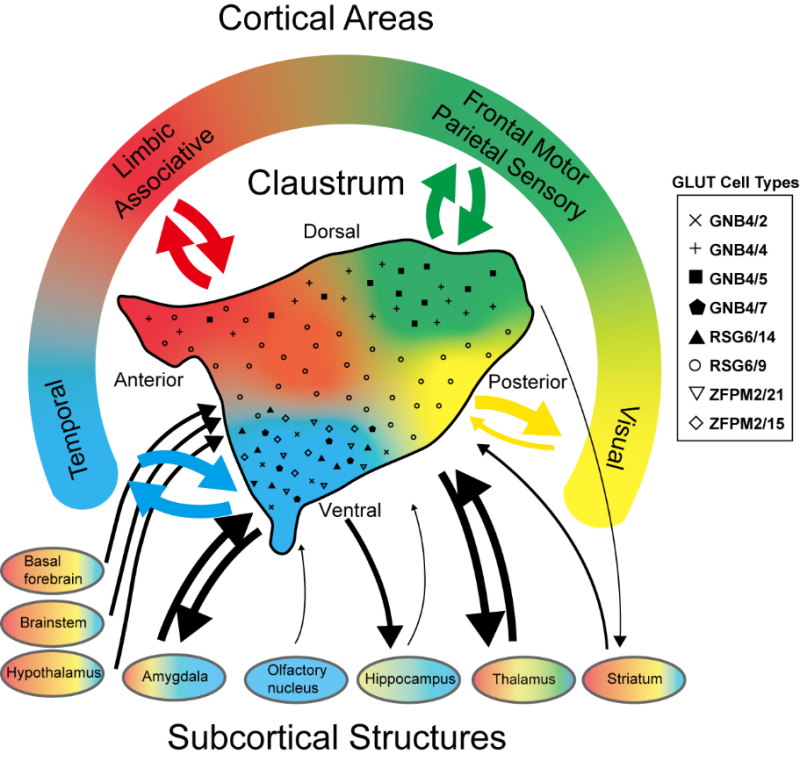 Schematic of four macaque claustrum domains with unique connectivity and cell profiles.
Schematic of four macaque claustrum domains with unique connectivity and cell profiles.
The research analyzed over 227,000 claustral cells from cynomolgus monkeys, identifying 48 distinct transcriptome-defined cell types, including 22 excitatory glutamatergic, 19 inhibitory GABAergic and seven non-neuronal cell types. Cross-region comparison reveal that more than 80 percent of the glutamatergic neurons closely resemble deep-layer insular neurons, lending strong support to the hypothesis of a shared developmental origin between the claustrum and the insular cortex.
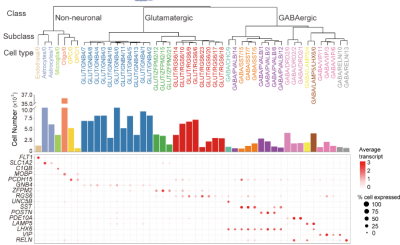
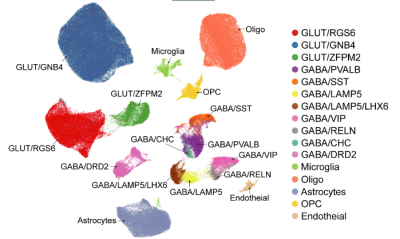
Single-nucleus RNA sequencing reveals 48 transcriptome-defined cell types in the macaque claustrum, grouped into three major classes and 16 subclasses.
.
The study further demonstrates that while the spatial distribution of most glutamatergic neurons is highly organized along medial-lateral and/or dorsal-ventral axes, the GABAergic neurons are distributed in a much more uniform pattern. Such spatial encoding not only underscores the functional compartmentalization within the claustrum but also hints at its pivotal role in integrating multimodal information across the brain.
The team utilized retrograde tracer injections at 171 strategically selected cortical and subcortical sites to map the brain-wide connectivity of the claustrum. This comprehensive connectivity analysis delineated four distinct projection-selective zones, each characterized by preferential connections with specific functional networks including prefrontal, visual, memory-related, and motor areas. By combining the connectivity data with detailed cell-type maps, the researchers were able to correlate specific molecular signatures with distinct projection patterns, suggesting that certain primate-specific glutamatergic cell types may underlie advanced cognitive functions such as abstract thinking and memory integration.
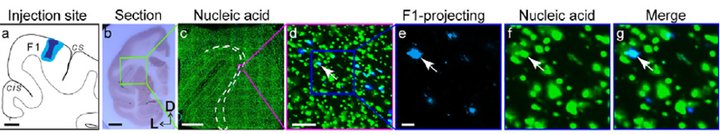 Combination of stereo-seq and retrograde labeling to match transcriptome-defined cell types with projection target-selective neurons.
Combination of stereo-seq and retrograde labeling to match transcriptome-defined cell types with projection target-selective neurons.
An important aspect of the study is its cross-species comparison, where the macaque data were integrated with similar datasets from marmosets and mice. This comparative analysis not only highlights the evolutionary divergence of the claustrum in primates, where cell types such as the GNB4 subtypes are enriched, but also underscores the unique adaptations that may contribute to the neural substrates of human consciousness. The discovery that primate claustral neurons are significantly more similar to deep-layer insular neurons than to those in other brain regions adds a new dimension to our understanding of the evolutionary trajectory of brain structures involved in high-order cognitive functions.
As part of a broader effort to decode the neural mechanisms underlying consciousness, the study provides an invaluable resource by making the full dataset publicly available at https://macaque.digital-brain.cn/claustrum. These findings open up new avenues for exploring how the interplay between distinct neural cell types and their connectivity patterns contributes to the emergence of consciousness and cognition in primates, potentially offering fresh perspectives on the evolution of the human brain.
 Website for macaque claustrum connectome and spatial transcriptomic atlas.
Website for macaque claustrum connectome and spatial transcriptomic atlas.
By uniting cutting-edge sequencing technologies with innovative circuit mapping, the BGI team has not only delivered a detailed molecular and connectivity blueprint of the macaque claustrum but has also provided compelling evidence for its role as a central hub in brain-wide information integration. The insights gleaned from this research hold promise for advancing our understanding of neurological disorders and could eventually inform novel therapeutic strategies aimed at modulating brain connectivity and cognitive processing.
Ethical approval for this study was obtained.
The research papers can be accessed here: https://www.cell.com/action/showPdf?pii=S0092-8674%2825%2900273-9



