Hemogen Therapeutic, a gene therapy company and subsidiary of BGI Group, in collaboration with Shenzhen Children’s Hospital, has published clinical research in the international, peer‑reviewed journal Molecular Therapy on hemobeglogene autotemcel (hemo-cel), an investigational gene therapy for transfusion-dependent β‑thalassemia (TDT).
 The study “Safety and efficacy of hemobeglogene autotemcel(hemo-cel) gene therapy in five patients with transfusion-dependent β-thalassemia” was published in Molecular Therapy.
The study “Safety and efficacy of hemobeglogene autotemcel(hemo-cel) gene therapy in five patients with transfusion-dependent β-thalassemia” was published in Molecular Therapy.
TDT can place a heavy burden on families. Many patients need regular blood transfusions for years, often starting in early childhood. Because frequent transfusions can cause iron to build up in the body, patients may also need long-term iron-removal treatment to help protect vital organs. While bone marrow transplantation can be curative for some patients, it depends on finding a suitable donor and carries significant risks. Hemogen Therapeutic is developing gene therapy as a different approach, designed to help patients produce functional hemoglobin using their own cells.
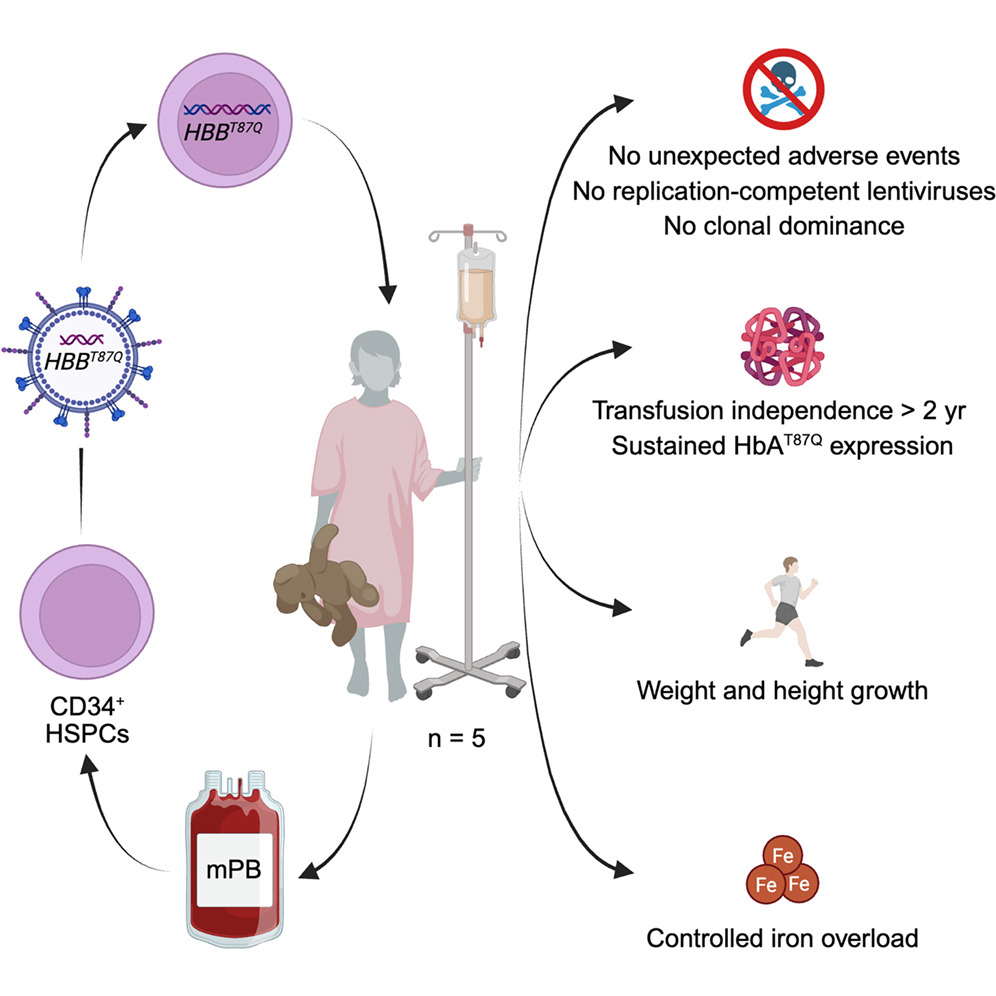
Five eligible participants, aged 8–16 years and diagnosed with TDT, were enrolled and treated at Shenzhen Children’s Hospital. In the study, patients received hemo-cel, an investigational treatment made from a patient’s own blood-forming stem cells. In the laboratory, these cells are modified using a lentiviral vector to add a functional β‑globin gene variant, with the goal of restoring the body’s ability to make effective hemoglobin and reduce or eliminate the need for transfusions.
 Graphical abstract
Graphical abstract
According to the paper, all five treated patients achieved transfusion independence, meaning they were able to stop regular red blood cell transfusions for an extended period after treatment while meeting the study’s hemoglobin criteria. At the time of analysis, each patient had maintained transfusion independence for more than two years, with a median transfusion-free period of 34.3 months. The paper also reports relatively rapid recovery of key blood counts after treatment, with neutrophil recovery at a median of 18 days and platelet recovery at a median of 13 days.
Safety was closely monitored, including specialized long-term risk assessments commonly used in lentiviral gene therapy. The publication reports that no replication-competent lentivirus was detected and that integration site analyses did not show evidence of clonal dominance during follow-up. The investigators reported that serious side effects were mainly associated with the conditioning chemotherapy (busulfan) used before infusion, and that no adverse events were considered related to the hemo-cel infusion itself.
The paper also describes broader health observations. During the reported 24-month follow-up window, patients did not resume iron chelation therapy, and the study describes findings consistent with stable or improved measures related to iron burden in evaluable patients, alongside gains in height and weight over time.
“This publication marks an important step for Hemogen Therapeutic as we continue to develop gene therapy options for patients who have historically faced lifelong treatment,” said Dr. Chao Liu, corresponding author and CEO at Hemogen Therapeutic. “We are grateful to our clinical partner Shenzhen Children’s Hospital and to the patients and families who participated. While this is an early-stage study with a small number of participants, the results provide encouraging clinical evidence that supports continued evaluation of hemo-cel.”
The clinical study was registered on ClinicalTrials.gov. The authors note that additional studies are underway to evaluate hemo-cel in broader patient populations and that long-term follow-up is planned to better understand safety and durability over time. The parents of all pediatric participants provided written informed consent, and the participants themselves gave written assent.
This study can be accessed here: https://doi.org/10.1016/j.ymthe.2026.02.017



