In Southeast Asia, where thalassemia remains highly prevalent, patients continue to face limited access to advanced gene therapy and insufficient localized manufacturing capacity.
In January 2026, Hemogen Therapeutic, a subsidiary of BGI Group, successfully completed the first overseas localized production of HGI-001 Injection (Hemo-cel) in collaboration with the Cell and Gene Therapy Manufacturing Center (CAGTMC) Laboratory at the Faculty of Medicine, Chulalongkorn University in Thailand. This achievement marks not only a significant milestone in Hemogen Therapeutic’s international technology transfer efforts, but also the first successful international validation of its SPARK global deployment model.
 Production and preparation of Hemo-cel at Chulalongkorn University in Bangkok, Thailand.
Production and preparation of Hemo-cel at Chulalongkorn University in Bangkok, Thailand.
SPARK is Hemogen Therapeutic’s integrated global deployment solution, developed to support international partners in rapidly establishing clinical-grade local gene therapy manufacturing capabilities.
The framework is designed to ensure the comprehensive implementation of technical systems, quality standards, and regulatory alignment - making localized thalassemia gene therapy more efficient, scalable, and accessible.
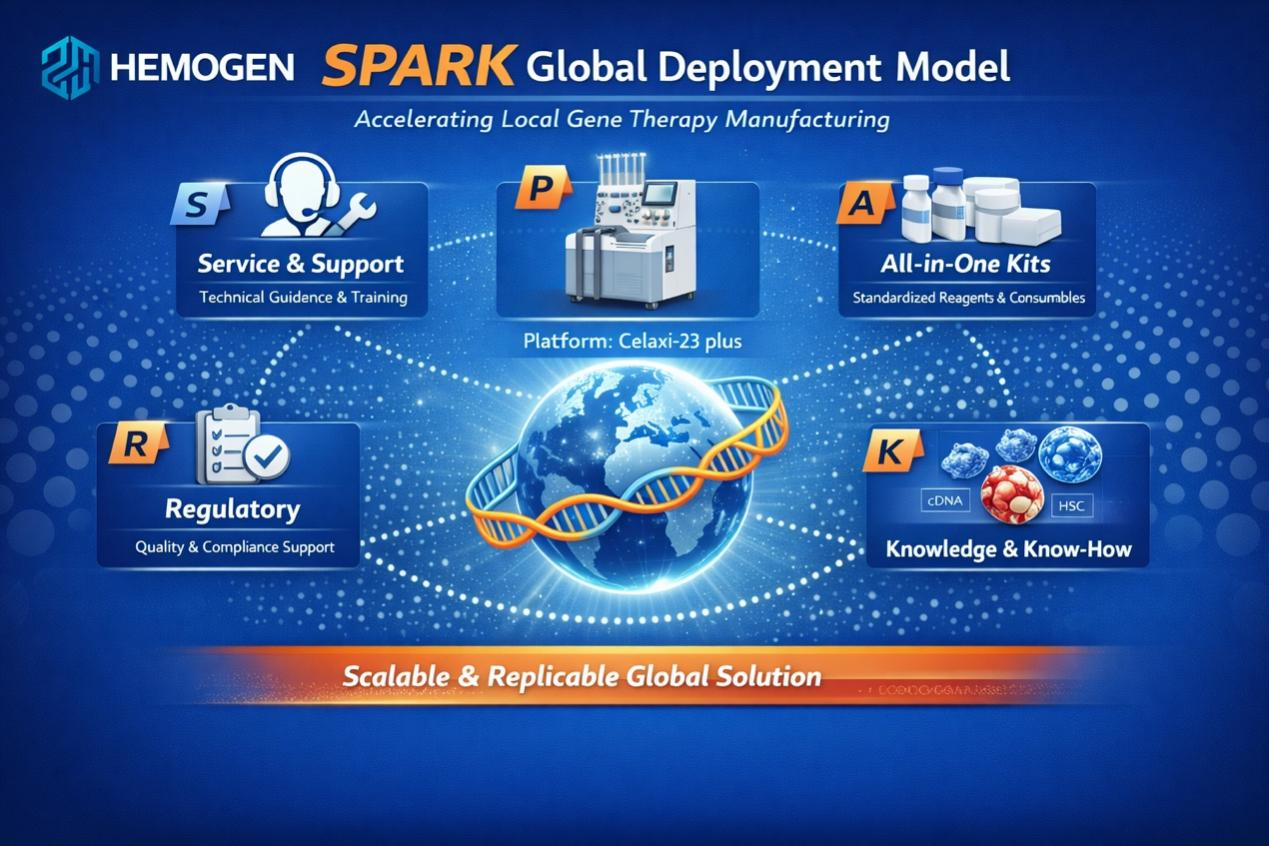 Hemogen Therapeutic’s SPARK global deployment Model
Hemogen Therapeutic’s SPARK global deployment Model
SPARK consists of five core pillars:
S – Service & Support: End-to-end technical guidance, on-site operational support, and structured training throughout the full implementation lifecycle to ensure stable deployment.
P – Platform: A fully integrated R&D-to-manufacturing platform anchored by Hemogen Therapeutic’s proprietary Celaxi-23 automated cell-processing system, enabling standardized and automated production.
A – All-in-One Kits: Standardized, ready-to-use manufacture kits and consumables designed to ensure reproducibility, consistency, and streamlined workflows across regions.
R – Regulatory: Comprehensive quality-system frameworks and regulatory documentation aligned with international standards to facilitate local registration and compliance.
K – Knowledge & Know-How: A structured technology transfer system enabling partners to internalize critical process parameters, operational expertise, and proprietary methodologies — ensuring long-term sustainability rather than one-time execution.
SPARK represents more than a technology package — it is a scalable and replicable global deployment model. The Thailand project serves as the first validated implementation of SPARK, demonstrating the successful transition of the framework from strategic design to real-world execution. This milestone establishes a foundation for broader regional expansion and improved accessibility to innovative thalassemia gene therapy across Southeast Asia.
Following the successful deployment in Thailand, Hemogen Therapeutic is actively extending the SPARK model to additional Southeast Asian countries, building a standardized regional manufacturing network.
At the same time, the company is expanding partnerships in the Middle East, where demand for cell and gene therapy (CGT) infrastructure and localized production capacity continues to grow rapidly.
Through SPARK, Hemogen Therapeutic aims to empower global partners to develop sustainable and scalable gene therapy ecosystems, accelerating long-term access to potentially curative treatments for inherited diseases worldwide.



