Every year on May 8th, the world observes International Thalassaemia Day to raise awareness of a condition that now knows no geographical boundaries. Thalassaemia has evolved into a global health concern with a significant burden: at least 1 in 20 people worldwide carries a haemoglobin disorder gene, and nearly 80% of affected births occur in low- and middle-income countries.
 Information Cards on the International Thalassaemia Day 2026 official website. (Credit: Thalassaemia International Federation)
Information Cards on the International Thalassaemia Day 2026 official website. (Credit: Thalassaemia International Federation)
Yet, despite decades of medical progress, it remains a largely "hidden" disease. Millions still live in the shadows, unaware of their carrier status—a silence that perpetuates suffering across generations. This year's theme, "Hidden No More: Finding the Undiagnosed. Supporting the Unseen," calls for urgent action on two interconnected fronts: finding the undiagnosed through early screening and providing comprehensive support for those already diagnosed but overlooked by health systems.
In many regions, the limitations of traditional diagnostic methods create significant gaps in screening infrastructure.
Conventional screening often requires multiple blood tests and repeated clinic visits—a time-consuming process that frequently misses opportunities for early diagnosis, particularly regarding rare mutations. In contrast, high-throughput sequencing (HTS) offers enhanced accuracy, higher sensitivity, and broader detection coverage. In high-prevalence regions, this approach will significantly improve early diagnosis rates and reduce misdiagnoses.
BGI Group applies HTS technology in thalassaemia screening test. Through BGI Genomics’ medical laboratory, specialized detection services provide a precise diagnostic foundation to reduce the incidence of severe cases. By integrating HTS and Gap-PCR, thalassaemia-seq assists doctors in identifying carrier status and assessing reproductive risks for couples, while facilitating timely infant diagnosis and intervention. As earlier screening could have made a profound difference, this approach aims to reach those previously overlooked.
For patients with severe thalassaemia, traditional treatment typically requires lifelong blood transfusions and chronic iron chelation. This regimen is not only costly but also severely diminishes the quality of life. While hematopoietic stem cell transplantation offers a potential cure, the difficulty of finding a matched donor means only a small minority of patients can access this option.
Addressing this critical need, Hemogen Therapeutic, a subsidiary of BGI Group, has developed Hemo-cel (HGI-001 Injection). This innovative gene therapy utilizes a lentiviral vector to deliver a functional β-globin gene into the patient's own hematopoietic stem cells, restoring the body’s ability to produce normal hemoglobin at the source.
 Production and preparation of Hemo-cel at Chulalongkorn University in Bangkok, Thailand.
Production and preparation of Hemo-cel at Chulalongkorn University in Bangkok, Thailand.
To ensure this life-changing therapy reaches those in need, in January 2026, Hemogen Therapeutic successfully completed the first overseas localized production of Hemo-cel with Cell and Gene Therapy Manufacturing Center (CAGTMC) Laboratory at the Faculty of Medicine, Chulalongkorn University in Thailand. This marks a significant milestone in international technology transfer and the first validation of its SPARK global deployment model. It enables international partners to rapidly establish clinical-grade local manufacturing. The success of the Thailand project demonstrates that advanced gene therapy no longer needs to be a distant luxury; instead, through localized production, it can become an efficient, scalable, and accessible reality for thalassaemia patients across Southeast Asia and beyond.
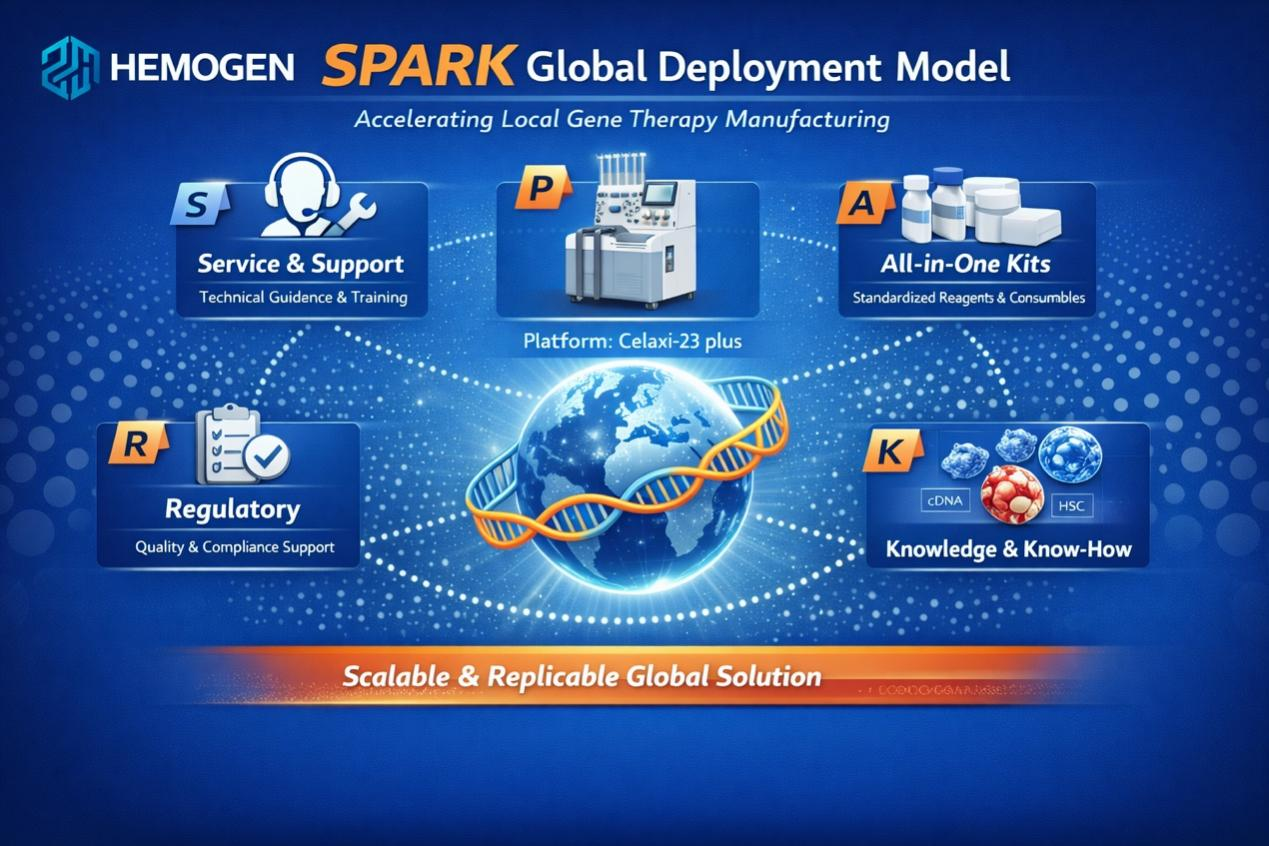 Hemogen Therapeutic’s SPARK global deployment model.
Hemogen Therapeutic’s SPARK global deployment model.
Alongside these efforts to improve the access to thalassaemia detection and treatment, BGI Group continues to provide direct humanitarian aid through its long-standing public welfare programs. As a co-founder of the Hua Foundation, BGI Group has been committed to providing free matching tests globally to patients with severe thalassaemia. Since 2017, the foundation has been offering free HLA matching for severe thalassaemia families nationwide. By February 2026, this initiative had benefited 7,930 families, with 862 patients successfully finding fully matched donors.
Recognizing that long-term success requires more than individual support, BGI Group also advocates for systematic, regional collaboration to build a sustainable prevention network. On November 24, 2025, a milestone cross-border public health collaboration—the Southeast Asia Thalassaemia Prevention Alliance (SEATPA)—was officially launched in Chiang Mai, Thailand. This historic step aims to unite countries, experts, and communities to strengthen control efforts, with the ultimate vision of eliminating preventable thalassaemia in the region. By fostering a multidisciplinary cross-border network, the alliance is dedicated to reducing disease incidence, improving patients' quality of life, and alleviating the economic burden on families and society.
 Official launch of SEATPA in Chiang Mai, Thailand, on November 24, 2025.
Official launch of SEATPA in Chiang Mai, Thailand, on November 24, 2025.
Thalassaemia will become "Hidden No More." The synergy of technological innovation, global strategic cooperation, and humanitarian care is breaking the silence that has persisted across generations. By ensuring every patient is seen and supported, the global community moves closer to a world where this disease no longer limits human potential. BGI Group remains dedicated to this commitment, continuing to provide innovative solutions and foster global partnerships to ensure a healthier future for all.



